 
For medical device developers, risks are a core principle of product development and should be tied together in one powerful platform. Risk management is an inextricable part of the medical device development process. Managing Risks & Requirements for ISO 14971 The standard was updated in 2019, providing more guidance on risk management and adding more detailed requirements. Jama Connect offers a straightforward approach to managing risk according to ISO 14971 in one platform. ISO 14971 is an international standard that sees risk management as a product lifecycle process encompassing the development, production, and post-production stages. 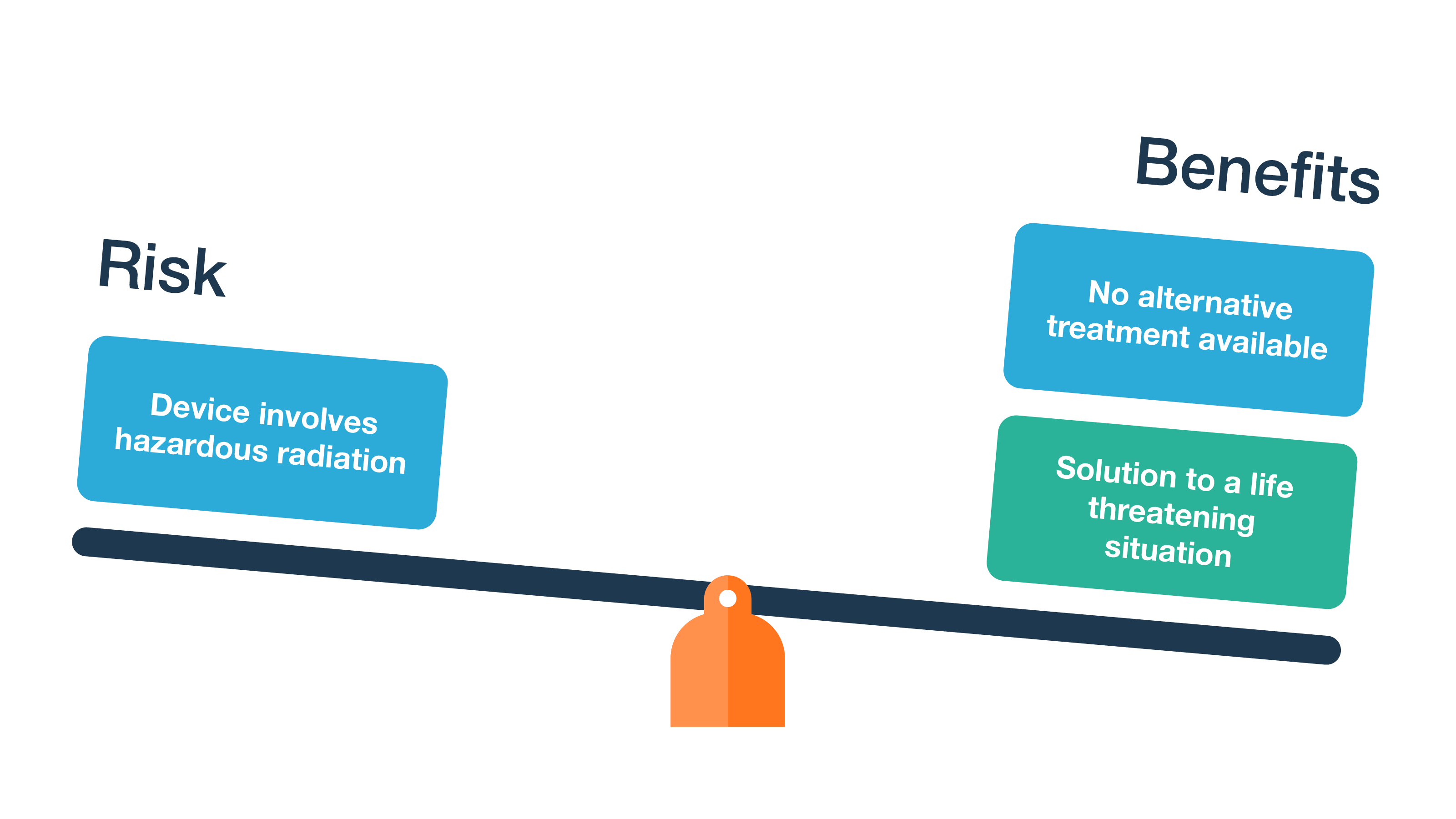 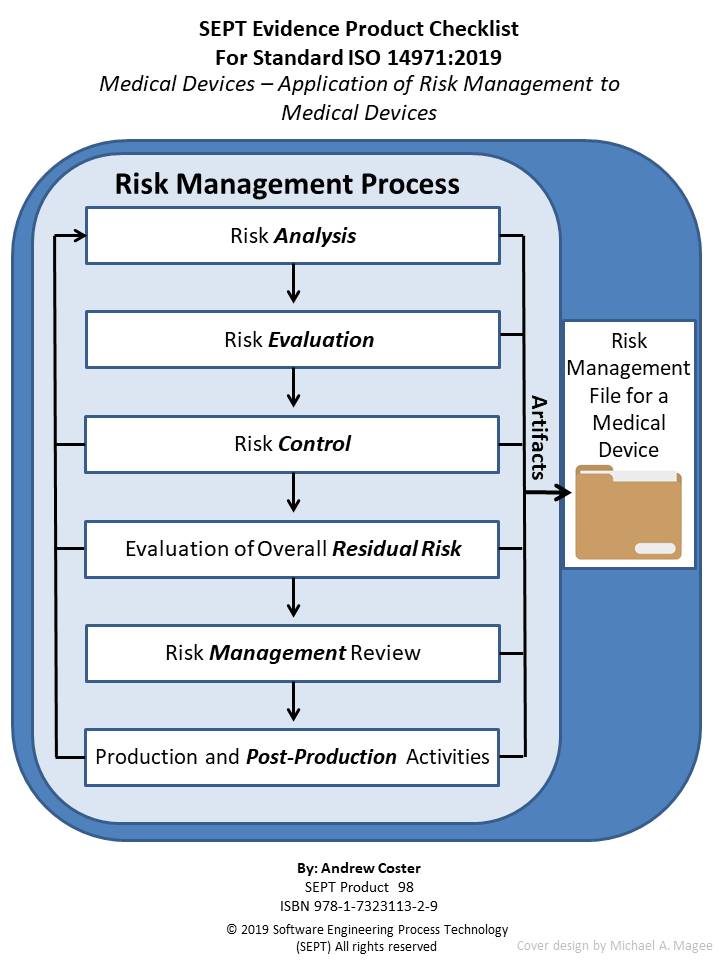
In this post, we’ll outline the main clauses of ISO 14971 and explain how Jama Connect can help medical device developers build better, safer products that satisfy ISO 14971. In our experience working with more than 200 medical device developers, we’ve realized how important it is to create best practices for risk management under ISO 14971, the FDA’s mandatory standard for risk assessment throughout the product development lifecycle. Last week, Jama Software launched Jama Connect® for Medical Device Development, which helps teams speed time-to-market without compromising quality or compliance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
